0+ Years
Experience

Beyond CRO.
Your Intelligent Clinical Partner.

We bridge clinical intelligence with biometrics to ensure data quality and accelerate timelines. As your intelligent clinical partner, we don’t just deliver data, we deliver certainty.
Experience
Studies Delivered
NDA/BLA Submission Approvals
Scalable, AI-enhanced biometrics solutions delivering submission-ready data with zero critical findings to accelerate your path to market.

With 1800+ studies across multiple therapeutic areas and 50+ NDA/BLA approvals, Q2BI delivers deep expertise that accelerates your path from trial to treatment.
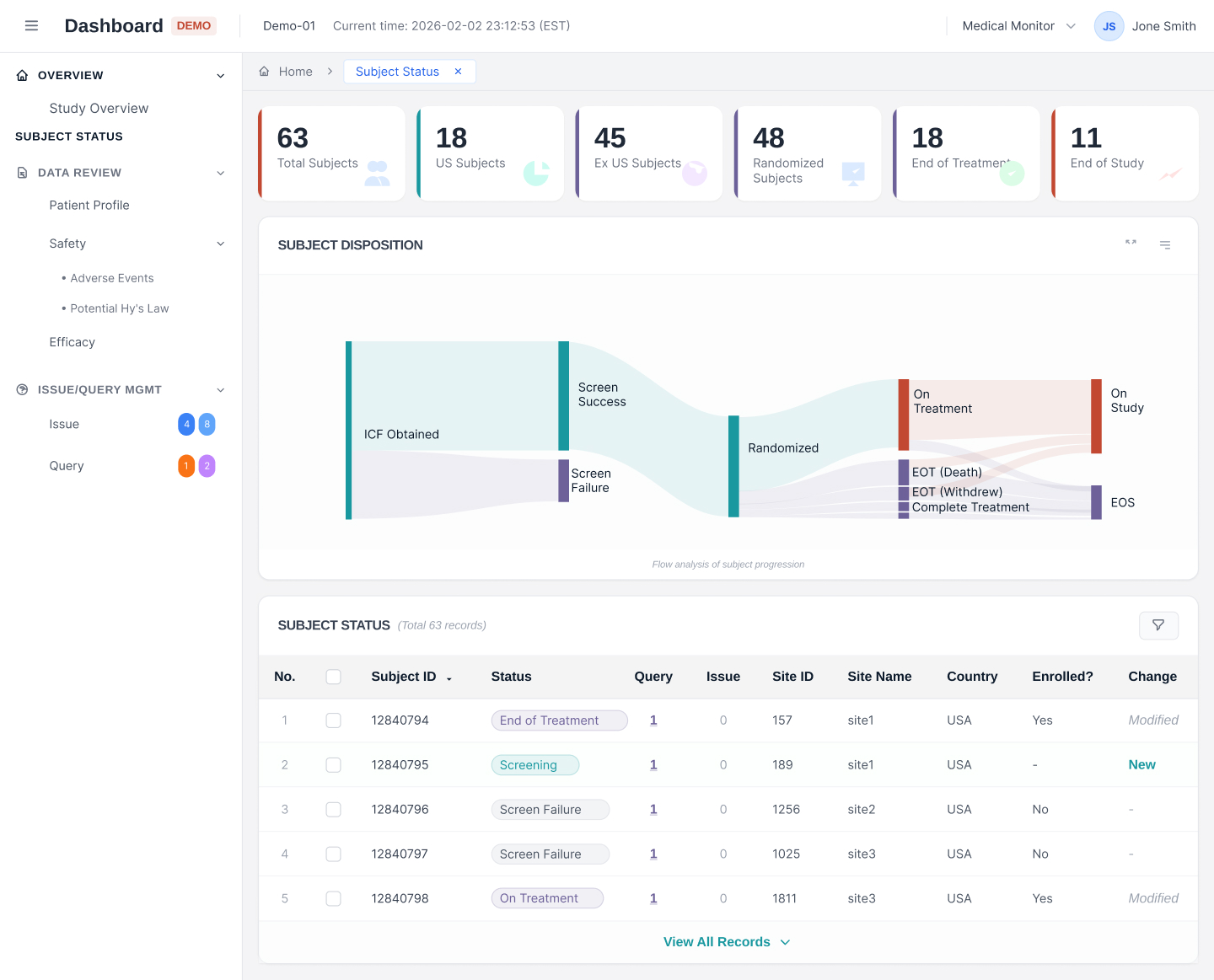
Incorporated automation processes in clinical trials to reduce repetitive tasks and catch issues early on.
Built an AI-powered visualization dashboard that helps track study progress and deliver actionable insights.
Developed distinct AI agents to help with image and data interpretation, as well as writing and research tasks.

We become an integrated extension of your team - not just a vendor, but a true partner committed to your success from first consultation to regulatory approval and beyond.
Seamless integration with your processes throughout the entire development lifecycle.
Real-time updates, proactive reporting, and open dialogue ensuring you are always informed.
Your success is our success - we take ownership and deliver results.
Tailored solutions, from full-service to FSP, designed for your unique needs.
Deep therapeutic expertise and regulatory knowledge driving quality outcomes.
AI-powered solutions and data intelligence accelerating your timelines.
Seamless integration with your processes throughout the entire development lifecycle.
Real-time updates, proactive reporting, and open dialogue ensuring you are always informed.
Your success is our success - we take ownership and deliver results.
Tailored solutions, from full-service to FSP, designed for your unique needs.
Deep therapeutic expertise and regulatory knowledge driving quality outcomes.
AI-powered solutions and data intelligence accelerating your timelines.
Stay updated on our latest breakthroughs, industry insights, and innovations accelerating the future of clinical research.

Featured Article
BOXBOROUGH, Mass. — March 24th, 2026 — Q-Square Business Intelligence, Corp. (Q2BI) today marked the independent launch of Janus Data Intelligence, Corp. (JDIX), a clinical AI technology company originally incubated with...
View DetailsYour Growth. Our Priority. Together We Bring Hope to Patients Faster.

Worksite: MA,NJ,CA
Worksite: MA,NJ,CA
Worksite: MA,NJ,CA
Worksite: MA,NJ
